Industrial Clean-in-Place (CIP) Unit for Pharmaceutical Processes
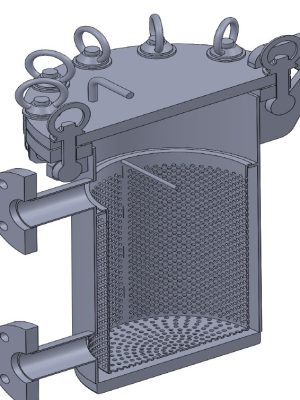
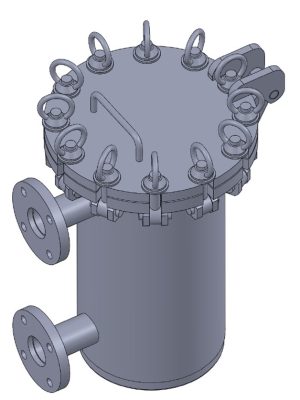
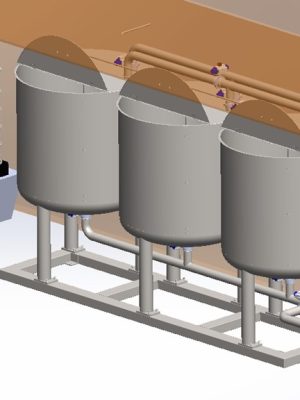
Detailed design and engineering of a compact three-stage Clean-in-Place (CIP) plant, configured for the automated sanitization of reactors and piping networks. The system allows for the sequential execution of initial wash, chemical descaling, and final rinse phases, guaranteeing the sterility standards required in pharmaceutical production.
Using SolidWorks, the 3D design of the unit was performed, optimizing the layout of the centrifugal pump, electrical heating elements, and the diaphragm pump system for descaling agent dosing. The control cabinet was equipped with high-precision instrumentation: Pt-100 temperature probes, pressure transducers, and conductivity meters. All detailed engineering, including dimensioning and construction plans for fabrication in pharmaceutical-grade stainless steel, was developed using AutoCAD.